Sponsors
-
Featured Companies See all (42)
-
Featured Conditions See all (31)
-
Preeclampsia
Gestational hyperten -
Narcotic addiction
Opioid Addiction -
Mononucleosis
Epstein–Barr v -
Morning Sickness
Hyperemesis gravidar -
Giardia
Giardiasis -
Post-Cholecystectomy
Post-Gallbladder Rem -
Megaloblastic anemia
Pernicious anemia -
Blastocystis hominis
Blastocystis hominis -
Gout
Podagra
-
Featured Nutraceuticals See all (50)
-
1,6-beta glucans
Glucans -
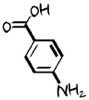
PABA
para-aminobenzoic ac -
Theanine
Gamma-glutamylethyla -
Vitamin B1
Thiamine -
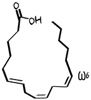
Gamma Linolenic acid
GLA, gamolenic a -
Apple Cider Vinegar
ACV, cider vineg -
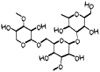
Arabinogalactan
Larch arabinogalacta -
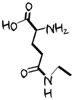
Taurine
Amino sulfonic acid -
Vitamin B7
Biotin -
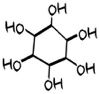
Inositol
Myo-inositol and D-c
-
-
Featured Herbals See all (46)
-
Gravel root; Joe pye
Eupatorium purpureum -
Cayenne
Capsicum annuum -
Chelidonium (Gre
Chelidonium majus -
Lily of the valley
Convallaria majalis -
Boldo
Peumus boldus -
Caterpillar fungus
Cordyceps sinensis -
Butcher's Broom
Ruscus aculeatus -
Eucalyptus
Eucalyptus globulus -
Aloe gel
Aloe vera -
Fennel
Foeniculum vulgare
-
-
Featured Actions See all (50)
-
Antitussive
Anticough -
Antiseptic
n/a -
Antirheumatic
Antiarthritic -
Diuretic
n/a -
Antihypertensive
Hypotensive